ABSTRACT
Objective: To evaluate the impact of the elexacaftor/tezacaftor/ivacaftor (ELX/TEZ/IVA) combination on the small airways in adults with cystic fibrosis (CF), a genetic disorder that primarily affects the respiratory system, leading to progressive lung disease. In CF, the small airways play a critical role, contributing to chronic symptoms such as cough, sputum production, and dyspnea. Methods: This was a single-center, retrospective observational study of adults with CF treated with ELX/TEZ/IVA for 12 months. We compared the patients who were homozygous for the F508del mutation of the CF transmembrane conductance regulator (CFTR) gene with those who were heterozygous for that mutation, in terms of lung function outcomes (FEV1, FEF25-75%, and the RV/TLC ratio) and the extent of non-homogeneous ground-glass opacity. Among the patients within the cohort, the same parameters were evaluated separately in those who had advanced lung disease and in those who had previously undergone CFTR modulator therapy. Results: There was a significant post-treatment improvement in lung function, with a median increase of 0.42 L/s in the FEF25-75% (p < 0.001) and a 5% reduction in the mean RV/TLC ratio (p < 0.001). There was a trend toward a higher improvement the F508del homozygous patients. A significant reduction in non-homogeneous ground-glass opacity was observed in 79.5% of the patients. Among the patients with advanced lung disease, there were notable post-treatment improvements in all of the parameters assessed. Conclusions: Our results highlight the positive impact that ELX/TEZ/IVA treatment can have on small airway function in patients with CF, with potential benefits even for those with advanced lung disease. Further research is needed in order to evaluate the long-term effects of this treatment and its relationship with patient-reported outcomes.
Keywords:
Cystic fibrosis/diagnostic imaging; Lung/physiopathology; Respiratory function tests; Lung/drug effects; Lung/diagnostic imaging; Cystic fibrosis/drug therapy.
INTRODUCTION Cystic fibrosis (CF) is a rare genetic disease with autosomal recessive inheritance, caused by variants in the CF transmembrane conductance regulator (CFTR) gene, that affects approximately 100,000 people worldwide.(1) Dysfunction of the CFTR protein leads to the development of multiple manifestations across various organ systems, with the respiratory system being the most prominent. Pulmonary involvement typically causes the most severe, progressive form of CF, ultimately leading to death. Over 2,000 variants have been described, the most common being F508del, which is found in nearly 90% of CF patients, of whom approximately 50% are homozygous for this mutation.(2)
The importance of the small airways in the pathophysiology of lung disease in CF is well known, because CFTR dysfunction causes viscous secretions and failure of mucociliary clearance due to respiratory mucin adherence to the airway epithelium. This leads to the production and accumulation of viscous bronchial secretions, air trapping, and increased respiratory muscle workload. Together, these factors contribute to chronic symptoms such as cough, sputum production, dyspnea, and reduced exercise capacity.(3)
In July of 2021, the CFTR modulator combination elexacaftor/tezacaftor/ivacaftor (ELX/TEZ/IVA) was approved for use in Portugal for patients who are homozygous for the F508del mutation and F508del heterozygous patients carrying a second minimal function mutation; in February of 2022, it was approved for those carrying at least one F508del allele.(4)
The safety and tolerability profile of ELX/TEZ/IVA have been well established, as have its benefits, including improvements in FEV1, a reduction in the number of pulmonary exacerbations, and a reduction in the need for inhaled antibiotics,(5) as well as enhanced quality of life, nutritional status, and survival.(6,7) However, there is still limited knowledge regarding the impact of this combination on the small airways.
In this study, we aimed to identify the functional and imaging outcomes of patients who initiated treatment with the ELX/TEZ/IVA combination, in order to assess its impact on small airways disease. We also aimed to compare the outcomes of ELX/TEZ/IVA treatment between F508del homozygous patients and F508del heterozygous patients.
METHODS This was a retrospective, observational, single-center study of adult CF patients followed at the Cystic Fibrosis Referral Center of the Hospital de Santa Maria, in the city of Lisbon, Portugal, who started treatment with ELX/TEZ/IVA and continued it for at least one year. Patient recruitment was conducted between September and November of 2024.
The collected data included age; sex; CFTR genotype; date of treatment initiation; prior treatment with other CFTR modulators; lung function, at baseline and after one year of treatment; and imaging findings on chest CT performed prior to the initiation of ELX/TEZ/IVA and after at least one year of treatment. Relevant medical histories were evaluated by reviewing patient medical records.
Lung function parameters were obtained through spirometry and plethysmography. Small airways function was assessed by measuring FEF25-75%, expressed in liters/second and as a percentage of the predicted value, calculating the RV/TLC ratio, and looking for a non-homogeneous ground-glass opacity pattern on chest CT. Improvement in the non-homogeneous ground-glass opacity pattern was subjectively assessed by a thoracic radiologist, who evaluated chest CT scans (performed during inspiration and expiration) and compared them to the previous scans. The impact that treatment with ELX/TEZ/IVA had on obstruction of the larger airways was also assessed by measuring FEV1, expressed in liters.
Because the study period overlapped with the transition to new Global Lung Function Initiative equations, some pulmonary function tests (PFTs) were conducted using the old equations and more recent PFTs were conducted using the new equations. Therefore, the analysis of the results focused on the comparison of absolute values, given that the use of different equations for predicting reference values could introduce bias. Advanced lung disease was defined as an FEV1 < 40% of the predicted value.
Lung function data were collected as close as possible to (before) the initiation of ELX/TEZ/IVA therapy (often on the same day). However, due to the constraints imposed by the COVID-19 pandemic, the median time from the pre-treatment lung function evaluation to the initiation of therapy was 82.55 days (IQR: 17.75-102.0 days). For the post-treatment evaluation, the PFT conducted soonest after at least 12 months of therapy was selected. The median time from the initiation of therapy to the follow-up PFT was 439.0 days (IQR: 385.3-491.8 days).
For descriptive statistics, continuous variables with normal distribution are presented as mean ± standard deviation, whereas the remaining data are expressed as median and interquartile range. Categorical variables are reported as absolute and relative frequencies. The mean differences between subgroups were analyzed by t-test for independent samples if the data had a normal distribution and by Mann-Whitney U test if the data had a non-normal distribution. Pre- and post-treatment comparisons of data with normal and non-normal data distribution were made with t-test for paired samples and the Wilcoxon test, respectively. Categorical variables were analyzed by using the chi-square test for independent comparisons and McNemar’s test for paired comparisons. Values of p < 0.05 were considered statistically significant. Statistical analysis was performed with the IBM SPSS Statistics software package, version 28.0 (IBM Corp., Armonk, NY, USA).
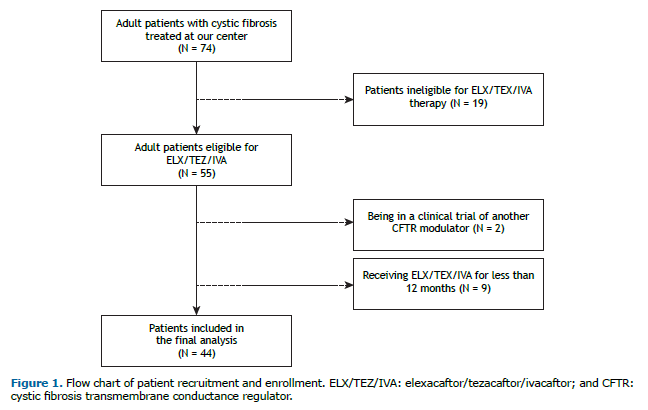
RESULTS During the study period, a total of 74 adult patients were followed at our referral center for CF. Of those, 55 were eligible for ELX/TEZ/IVA therapy, in agreement with national recommendations. Nine patients were excluded because they had been on the therapy for less than 12 months and 2 because they were on a clinical trial of another CFTR modulator.
Patient recruitment, enrollment, and follow-up are depicted in Figure 1. Table 1 summarizes the demographic and clinical features of the study cohort at baseline. The mean age at the diagnosis of CF was 13.4 ± 12.5 years. The mean age at initiation of ELX/TEZ/IVA therapy was 31.9 ± 12.5 years. Advanced lung disease was seen in 8 (18.2%) of the 44 patients.
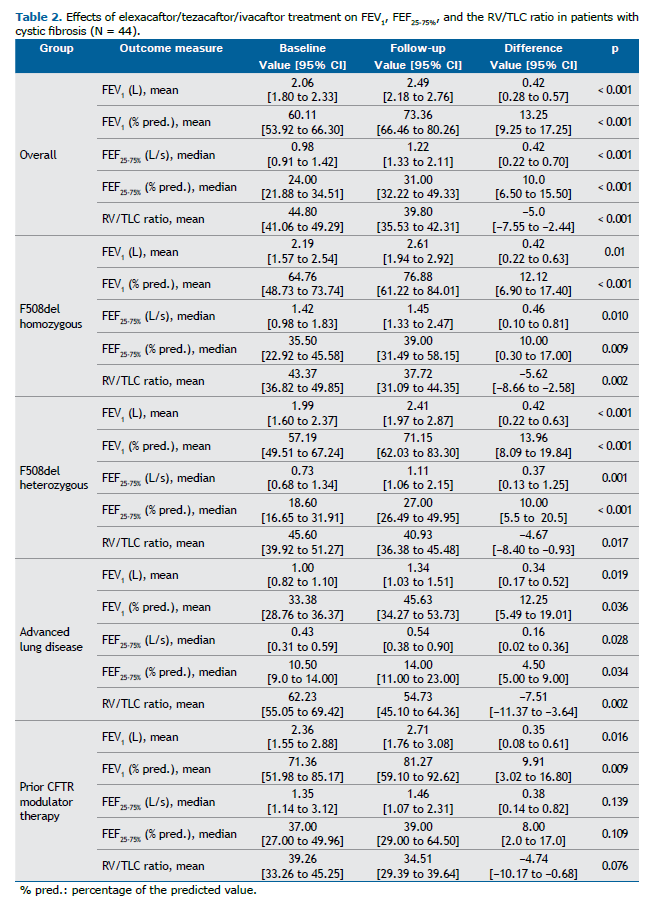
After 12 months of ELX/TEZ/IVA therapy, there was a median increase of 0.42 L/s in the FEF25-75% (p < 0.001) and a 5% reduction in the mean RV/TLC ratio (p < 0.001). A post-treatment improvement in the non-homogeneous ground-glass opacity pattern was observed in 35 (79.5%) of the 44 patients evaluated. The median time from the initiation of therapy to the follow-up chest CT was 623.0 days (IQR: 424.3-795.0 days). Of the 17 patients in the F508del homozygous group, 14 (82.4%) showed improvement in that pattern, compared with 77.8% of the 27 patients in the heterozygous group. Even among the 8 patients with advanced lung disease, 7 (87.5%) exhibited radiological improvement in the pattern. Among the 11 patients previously treated with other CFTR modulators, 8 (72.7%) showed radiological improvement in the non-homogeneous ground-glass opacity pattern.
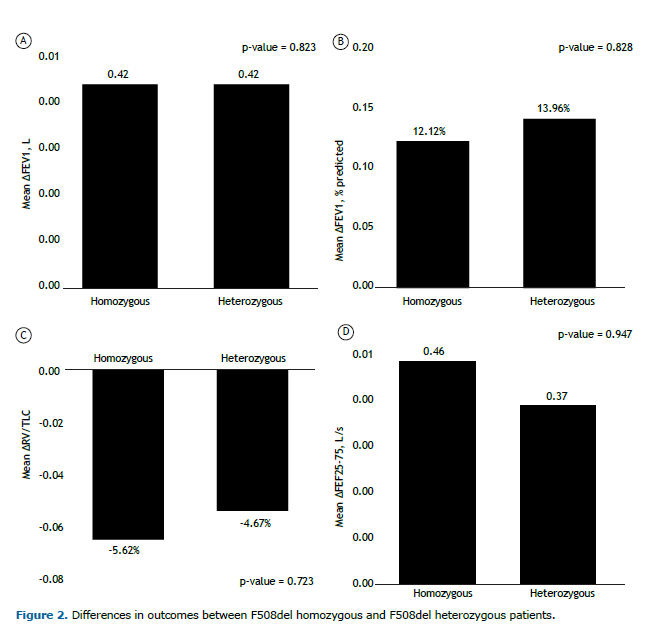
The degree of improvement in FEV1, as an absolute value and as a percentage of the predicted value, was similar between the F508del homozygous patients and the F508del heterozygous patients (Table 2). As shown in Figure 2, the improvements in the small airways, as assessed by measuring the FEF25-75% (in L/s) and the RV/TLC ratio, were greater in the F508del homozygous patients, although the differences in comparison with the F508del heterozygous patients were not statistically significant (p = 0.947 and p = 0.723, respectively).
DISCUSSION The benefits of highly effective CFTR modulator therapy for lung function, particularly for improving FEV1, are well-established in the literature.(7-10) To our knowledge, data on the impact of such therapies on the small airways are sparse. In this real-world study, we have provided clear evidence of the positive impact that 12 months of treatment with ELX/TEZ/IVA has on the small airways, observing a median improvement of 0.42 L/s in the FEF25-75% and a 5% reduction in the mean RV/TLC ratio. When evaluating the impact on patients with advanced lung disease, we observed statistically significant improvements across all parameters assessed. The greatest improvement was seen in the RV/TLC ratio, whereas the improvement in the FEF25-75%, albeit statistically significant, was more modest.
Although trends in improvement in the small airways were observed in patients previously treated with other CFTR modulators, those changes were not statistically significant. That might reflect a gain achieved with previous treatment, rendering further improvement more challenging to assess.
We also observed improvements in the non-homogeneous ground-glass opacity pattern on chest CT, in homozygous and heterozygous patients, as well as in those previously treated with other CFTR modulators, and even in patients with advanced lung disease.
These results highlight the potential of ELX/TEZ/IVA therapy to target small airway dysfunction, which is critical in the management of CF. Further studies are needed in order to assess the long-term effects of this therapy on the small airways, as well as its relationship with patient-reported outcomes.
AUTHOR CONTRIBUTIONS SCS and EF: patient follow-up; data collection; statistical analysis; and drafting of the manuscript. AB and MC: patient follow-up; and data collection. PA: study conception and design; and patient follow-up. All the authors revised the manuscript, approved the final version as submitted, and agree to be accountable for all aspects of the work.
CONFLICTS OF INTEREST EF receives speaking and lecture fees from Vertex Pharmaceuticals, Inc. PA receives consulting or advisory fees, as well as speaking and lecture fees, from Vertex Pharmaceuticals, Inc.
REFERENCES 1. Guo J, Garratt A, Hill A, Worldwide rates of diagnosis and effective treatment for cystic fibrosis. J Cyst Fibros. 2022;21(3):456-462. https://doi.org/10.1016/j.jcf.2022.01.009
2. Elborn JS. Cystic fibrosis. Lancet. 2016; 388(10059):2519-2531. https://doi.org/10.1016/S0140-6736(16)00576-6
3. Davis S D, Brody AS, Ratjen F. Cystic Fibrosis. In: Grippi MA, Elias JA, Fishman JA, Kotloff RM, Pack AI, Senior RM, editors. Fishman’s Pulmonary Diseases and Disorders. 6th ed. New York: McGraw Hill; 2022. p. 815-835
4. República Portuguesa. Serviço Nacional de Saúde, Autoridade Nacional do Medicamento e Produtos de Saúde (Infarmed) [homepage on the Internet] Lisboa: Infarmed; c2020 [updated 2022 Feb 21; cited 2024 Nov 08]. Relatório de avaliação de financiamento público de Medicamento Kaftrio (elexacaftor-tezacaftor-ivacaftor) + Kalydeco (ivacaftor). [Adobe Acrobat document, 25p.]. Available from: https://tinyurl.com/58s4nadu
5. Keogh RH, Cosgriff R, Stephenson A, et al. Real-world longitudinal outcomes of elexacaftor/tezacaftor/ivacaftor in cystic fibrosis. Respir Med. 2024;107882. https://doi.org/10.1016/j.rmed.2024.107882
6. Heijerman HGM, McKone EF, Downey DG, Van Braeckel E, Rowe SM, Tullis E, et al. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: a double-blind, randomised, phase 3 trial. Lancet. 2019;394(10212):1940-1948. https://doi.org/10.1056/NEJMoa1908639
7. Middleton PG, Mall MA, Dřevínek P, Lands LC, McKone EF, Polineni D, et al. Elexacaftor-Tezacaftor-Ivacaftor for Cystic Fibrosis with a Single Phe508del Allele. N Engl J Med. 2019;381(19):1809-1819. https://doi.org/10.1056/NEJMoa1908639
8. Sutharsan S, Dillenhoefer S, Welsner M, Stehling F, Brinkmann F, Burkhart M, et al. Impact of elexacaftor/tezacaftor/ivacaftor on lung function, nutritional status, pulmonary exacerbation frequency and sweat chloride in people with cystic fibrosis: real-world evidence from the German CF Registry. Lancet Reg Health Eur. 2023;32:100690. https://doi.org/10.1016/j.lanepe.2023.100690
9. Nichols DP, Paynter AC, Heltshe SL, Donaldson H, Frederick CA, Freedman SD, et al., Clinical effectiveness of elexacaftor/tezacaftor/ivacaftor in people with cystic fibrosis: a clinical trial, Am. J. Respir. Crit. Care Med. 205(5):529-539. https://doi.org/10.1164/rccm.202108-1986OC
10. Griese M, Costa S, Linnemann RW, Mall MA, McKone EF, Polineni D, et al., Safety and Efficacy of Elexacaftor/Tezacaftor/Ivacaftor for 24 Weeks or Longer in People with Cystic Fibrosis and One or More F508del Alleles: Inter-im Results of an Open-Label Phase 3 Clinical Trial, Am J Respir Crit Care Med. 203(3):381-385. https://doi.org/10.1164/rccm.202008-3176LE




 English PDF
English PDF
 Print
Print
 Send this article by email
Send this article by email
 How to cite this article
How to cite this article
 Submit a comment
Submit a comment
 Mendeley
Mendeley
 Pocket
Pocket